Yesterday, I was watching a video where the teacher had asked his students a question. He had just discussed how penicillins and cephalosporins can be used against certain bacteria, right after talking about Mycoplasma.
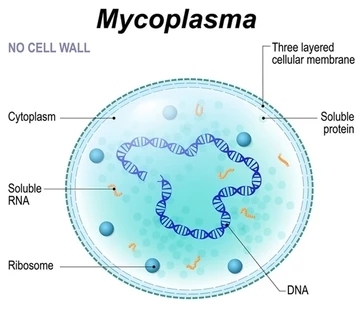
The teacher then asked the class: “Which is Mycoplasma more sensitive to: penicillins or cephalosporins?” One student raised his hand and answered, “Penicillin”. To this, the teacher replied, “That is wrong.” Immediately, the teacher asked another student, “So, what do you think Mycoplasma is more sensitive to: penicillins or cephalosporins?” Seeing that the first student was wrong when he had answered the question, the second student was perhaps quite confident that he would get it correct if he answered cephalosporins, and that was exactly what the second student did. Unfortunately, he was wrong! I absolutely saw it coming though, as this teacher is known to ask such trick questions to test students’ concepts. The teacher was visibly lamenting that they both got it wrong.
This was obviously a trick question. Mycoplasma has an equal sensitivity (better seen as an equal lack of sensitivity) to both penicillin and cephalosporins because while both classes of drugs work on bacterial cell walls, it has to be understood that Mycoplasma does not have a cell wall, and specifically, a cell wall with peptidoglycan! Consider this similar scenario. A person has no ears, and cannot effectively listen or hear anything. Which would give him a more enjoyable music-listening experience: earbuds or headphones? Hahaha!
The teacher’s original question is good because it reinforces the concepts which students are taught. This is how students actively learn: by seeing how the dots connect. A student can learn these three things separately: 1) the mechanism of action of cephalosporins; 2) the mechanism of action of penicillins; 3) the characteristics of Mycoplasma. Questions like the one asked by the teacher are effective in getting students to integrate the knowledge that they have learnt separately, at least from anecdotal experience.
As of the time of writing, Mycoplasma is arguably the smallest known living organism. What defines life? What defines a living organism? While there isn’t a unanimous consensus within the scientific community regarding the definition of life, a majority within the scientific community agree that prions, plasmids, transposons, bacteriophages, viruses all do not constitute life. As such, bacteria would generally constitute the smallest and “simplest” life forms. They are prokaryotes and are unicellular.
Most bacteria have cell walls. Why do they need a cell wall? Well, they need cell walls to prevent osmotic burst as their cell membranes are unstable due to a lack of sterols, which confer stability to the cell membrane. Recall that the cell membrane consists of mostly consists a lipid bilayer. The difference between a phospholipid and a triglyceride is that instead of having 3 fatty acid chains attached to a glycerol backbone in triacylglycerols, there are only 2 fatty acid chains attached to a glycerol backbone in phospholipids. A polar negatively-charged phosphate group takes the place of where the third fatty acid would have been connected. As such, the phosphate group, along with the carboxylic esters formed between the glycerol backbone and the two fatty acids constitute the hydrophilic head of the phospholipid while the tails of the two fatty acid constitute the hydrophobic tails of the phospholipid. What I have explained is beautifully illustrated in the diagram below.

With the hydrophilic heads of phospholipids facing the extracellular fluid and the cytosol, polar and charged substances are not able to diffuse through the cell membrane. However, despite being a polar molecule, water is able to diffuse through the cell membrane because it is a very small molecule.
As such, if not for the cell wall, bacteria would undergo osmotic burst or cytolysis due to excess water diffusing into the cell (we have just previously established that water is able to diffuse through the lipid bilayer despite being a polar molecule), and that the cell membrane is not large enough to accommodate the excess fluid.
This is where the cell wall comes in to help the bacteria resist cytolysis. But before we discuss how the cell wall, let us first discuss which living organisms have cell walls and their variations.
For the sake of our discussion, let us ignore plants and non-infectious agents since I am interested in infectious agents. For eukaryotic infectious agents, we have fungi, protozoa, helminths, and ectoparasites. Of these eukaryotic infectious agents, only fungi have cell walls. As for prokaryotes, we basically only have bacteria, and most bacteria have cell walls. Here is a diagram showing the structure of a fungi cell wall.
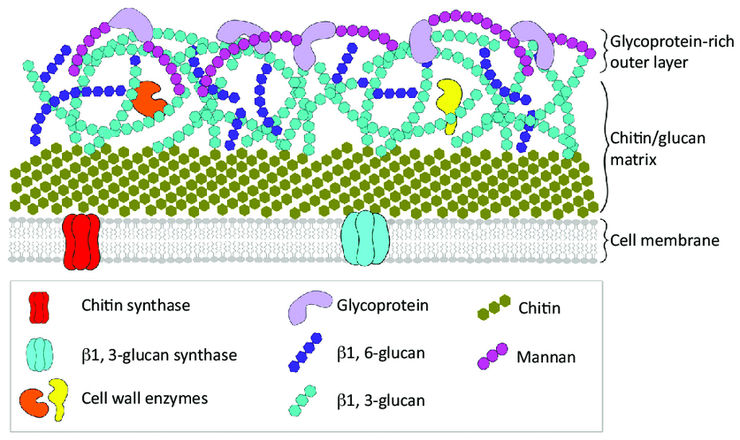
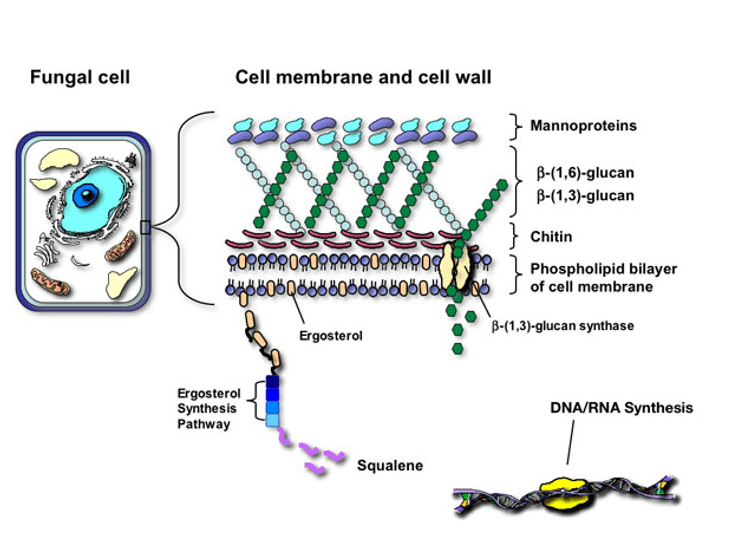
The difference between the cell walls in fungi and in bacteria is in the composition. Fungi cell walls consist of chitin, a polymer of N-acetylglucosamine, which is a glucose derivative. On the other hand, bacteria cell walls consist of peptidoglycan. The first step to understanding peptidoglycan is simply looking at its name. Consider “peptido-” and “-glycan”. This tells us that peptide bonds and sugar or sugar derivatives are involved in peptidoglycan’s structure. Consider this excellent diagram below which illustrates the structure of peptidoglycan beautifully:
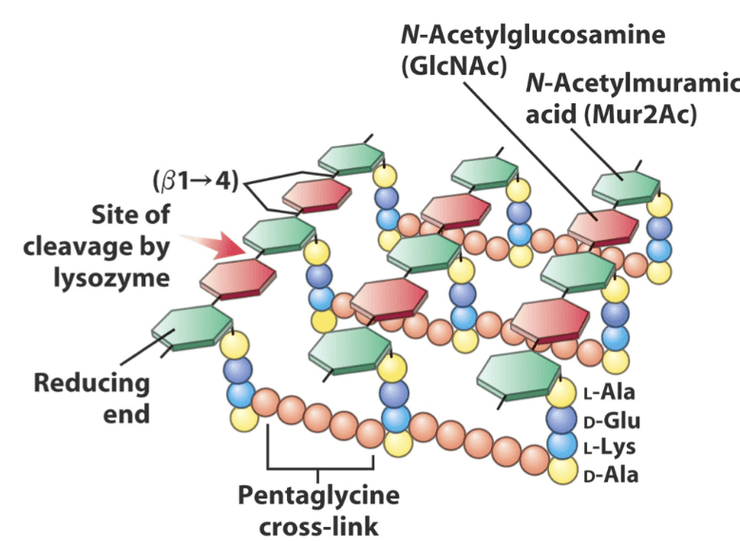
With a cell wall, bacteria are able to resist cytolysis.
The answer is that Mycoplasma has sterols within its cell membrane for stability. This presence of sterols within the cell membrane is unique among prokaryotes and it strongly suggests that sterols function as regulators of membrane fluidity.
The most well-known sterol that probably all of us have heard of is none other than cholesterol. The prefix “chole-” means bile or gall, which is a fluid produced in the liver. Bile consists mainly of water (97%), bile salts (0.7%), other substances, and of course, cholesterol.
Let’s take a look at the diagram involving peptidoglycan again. Do you see those pentaglycine cross-links? As the term suggests, these cross-links consist of 5 (penta) glycine residues linked together, with one terminal glycine joined to L-Lysine while another terminal glycine is joined to D-Alanine.
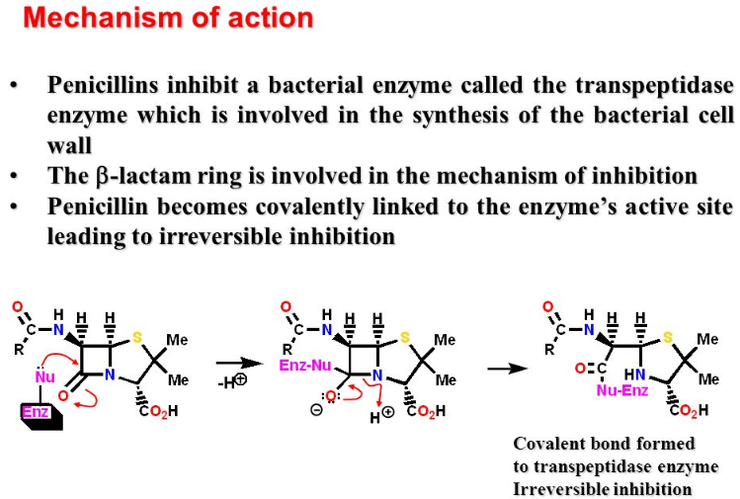
Penicillin works by binding to penicillin-binding proteins, which ultimately leads to cytolysis. One such protein is transpeptidase, the enzyme which catalyses the pentaglycine cross-links that connect the amino group on one N-Acetylmuramic acid to the amino group on another N-Acetylmuramic acid. When penicillin binds to transpeptidase, penicillin inhibits transpeptidase.
As such, with penicillin, these pentaglycine cross-links do not form, and the cell wall is not as sturdy as it should be, causing the bacterium to be more susceptible to cytolysis.
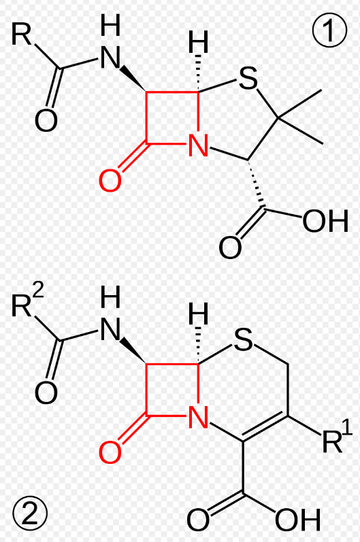
In the above image, we have the core structures of penicillin above (1), and the cephalosporins below (2). The part highlighted in red is known as the beta-lactam ring. It is named as such because a lactam is a cyclic amide (which is evident from C=O and the C-N bonds within the red 4-membered ring), and it is a beta-lactam because if the peptide bond were hydrolysed, then the nitrogen atom would be attached to the beta-carbon. The takeaway is that both penicillins and cephalosporins both belong to a broader category of antibiotics known as beta-lactams.

In all 20 standard naturally occuring amino acids, the amino group is bonded to the alpha-carbon, which is defined as the carbon immediately adjacent to the carboxylic acid group. It then follows that the beta-carbon would be immediately adjacent to the alpha-carbon and so on.
NB: This is why the most common ketone body in our bodies, 3-hydroxybutyrate, is also called beta-hydroxybutyrate (not technically a ketone). In beta-hydroxybutyrate, the hydroxyl group is attached to the beta-carbon. This is also why mitochondrial lipolysis is also known as beta-oxidation.
Some bacteria contain enzymes known as beta-lactamases, and as its name suggests, they degrade and thus inactivate beta-lactams. Such enzymes can be thought of as bacteria’s defence against our antibiotics. Bacteria which are capable of producing beta-lactamases include Staphylococcus, Neisseria gonorrheae, Mycobacterium tuberculosis, etc.

In response, to these beta-lactamases, we stubborn human beings who refuse to succumb to bacteria have developed what is known as beta-lactamase inhibitors. This is our strategy of combating the resistance put forth by the bacteria in the form beta-lactamases which break down our beta-lactam antibiotics. So, we developed a weapon against their weapon which worked against our initial weapon.
It wouldn’t be surprising if bacteria develop an enzyme which inactivates our synthetic beta-lactamase inhibitors. Then perhaps, we need to come up with something called the beta-lactamase inhibitor-inactivator inhibitor. This is a vicious cycle that may just carry on: beta-lactamase (inhibitor-inactivator) (inhibitor-inactivator) (inhibitor-inactivator) (inhibitor-inactivator) ad infinitum!
Yours faithfully,
Nic Loh
10 July 2019
References & Further Reading
1. https://www.sciencedirect.com/science/article/pii/S0070216108603111
2. https://en.wikipedia.org/wiki/Bile
3. https://www.sciencedirect.com/topics/immunology-and-microbiology/penicillin-binding-proteins

Leave a comment