NOTE: To make this blog post more educational, I have decided to come up with some questions throughout this blog post which you can contemplate to reinforce your understanding. My suggested answers can be found at the bottom of the post, just above the references. Feel free to give me feedback so that I can improve these questions and/or answers.
—-
For a long time, I thought that the (-) in blood type labels represents the lack of an antigen while the (+) represents the presence of that same antigen. I thought that the Rh system worked just like the ABO blood group system. So, I had always thought that the truly universal blood was O(-), and not O(+). My understanding was incomplete as the ABO system and the Rh system are not that similar. Even within the ABO system, O is not truly universal as individuals of the Bombay blood type cannot receive type O blood. Surprised? Allow me to explain.
There are many red blood cells (RBCs) in our body. These red blood cells are also called erythrocytes (“erythro-” comes from the Ancient Greek word ἐρυθρός or ‘eruthros’ which means ‘red’; “-cyte” comes from the Latin cyta, which in turn comes from Ancient Greek word κύτος or kutos which means ‘vessel or jar’). In most scenarios, the suffix “-cyte” just means “cell”.
NB: Not all substances with the -cyte suffix are cells. For example, platelets are called thrombocytes. Just looking at prefix and suffix of this word suggests that platelets are cells that lead to the formation of a thrombus. This is only partially right because while platelets help in the formation of a thrombus, platelets are not cells. Platelets are large fragments which are produced and released by megakaryocytes, which are basically large cells in the bone marrow. Platelets are not considered cells because they lack a nucleus and hence, lack nuclear genes. They do however have mitochondrial DNA, and nearly every other feature of a normal cell.
All of us have heard of blood donation. Some of us have even gone for blood tests or donated blood. The first time I went for a blood donation drive, all was fine until the last moment, where the volunteer was unable to find a suitable vein to draw blood from. Such an anticlimax. How does this all work? Historically, when blood transfusions were first attempted, immediate or delayed agglutination and subsequent hemolysis often occurred, and this often led to death.
As you might imagine, doctors were curious as to why this was happening. It was soon discovered that the bloods of different people have different antigenic and immune properties. Hence, what happened in those adverse transfusion cases was that the agglutinins present in the plasma of one person binded with the agglutinogens present on the RBCs of a different blood type. The antigens were treated as foreign invaders and this triggered a severe immune reaction. Fortunately, what this also meant was that if one can predict ahead of time the blood types of individuals, one could also thus predict if an adverse reaction would occur.
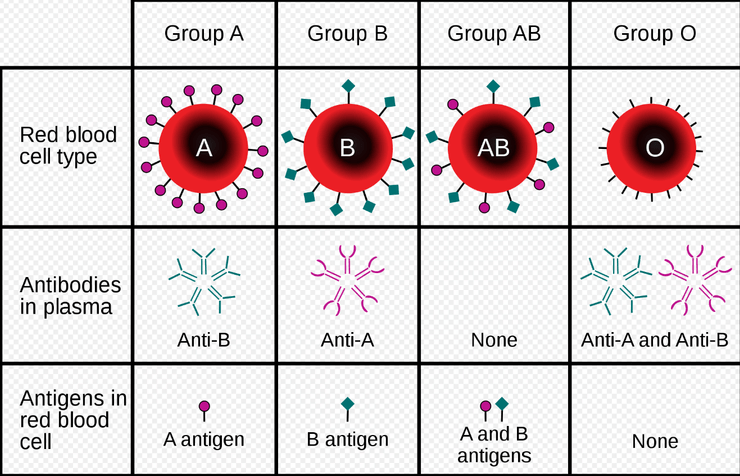
It should also be said that there are many other types of antigens on the outer surface of RBCs (about 30 which are common and hundreds of other rare antigens), which can all trigger antigen-antibody reactions. However, most of these reactions are weak. So, these other antigens are mainly used for studying the inheritance of genes to establish genealogy. Two particular types of antigens are much more likely to cause blood transfusion reactions, so we will be discussing those two types today: 1) the O-A-B system of antigens, and 2) the Rh system.
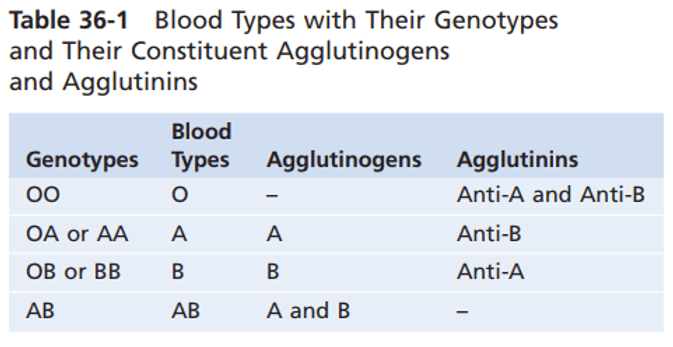
Firstly, within the O-A-B system, people typically have one of four blood types: O, A, B, AB. Whether one’s blood type is O, A, B, or AB is determined by the presence or absence of the two agglutinogens: the A and B agglutinogens, which are glycoproteins. What this means is that if your RBCs have neither the A or B, you are blood type O. If you have the A but no B, you are blood type A. If you have the B but no A, you are blood type B. If you have both the A and B, then you are type AB.
The type of agglutinogen you have depends on your genes. The genetic locus for the ABO blood group has three alleles (i.e. three variations of the same gene). For simplicity’s sake, we’ll call the alleles A, B, and O. To be extremely precise, Allele O technically results in the production of agglutinogen H, which is the precursor for for agglutinogens A and B (I’ll talk more about this antigen H later; for now just know that antigen H is also expressed on people blood types A, B, and AB). The diagram below is an excellent visual aid. You can clearly see that to get antigen A from antigen H, N-acetylgalactosamine needs to be added to the distal galactose group on antigen H. You can also see that to get antigen B from antigen H, a galactose group is added to the distal galactose group on antigen H.
Antigen h forms instead of Antigen H when the terminal fucose group is missing. People who lack antigen H and have antigen h instead are known to have the Bombay blood type or hh blood type. But of course, you will almost never hear of this Bombay blood type being mentioned online or in any textbook because of its sheer rarity (4 per million worldwide). Hence, even the production of the H antigen itself is often ignored because it is often deemed the baseline which almost everyone already has.
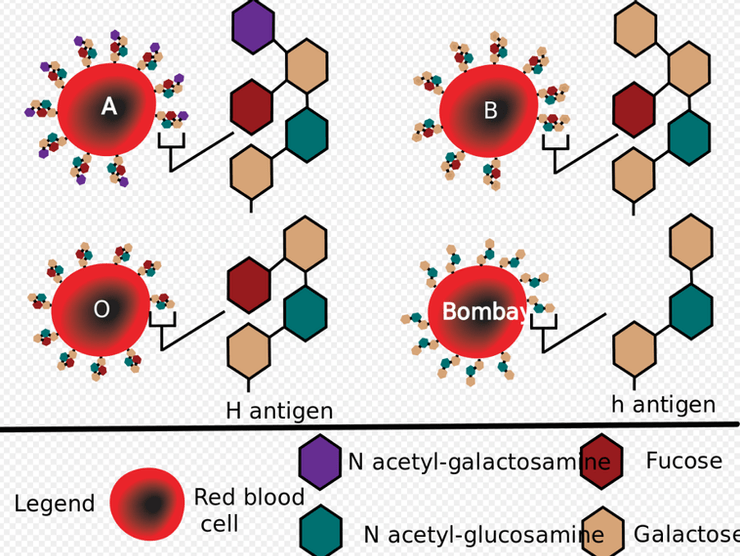
On the other hand, the alleles A and B result in strong agglutinogens on the cells. Thus, the O allele is recessive to both the A and B alleles, both of which demonstrate codominance.
However, each person only has two sets of chromosomes (one set from your mother and one set from your father). While a person with blood type AB may have all three antigens A, B, H, he can only have a maximum of two of the three alleles (i.e. A and B). Since there are 3 different alleles and 2 chromosomes, then 3 * 2 = 6 genotypes are possible: OO, OA, AA, OB, BB, AB. OO will give rise to type O blood, OA and AA will give rise to type A blood, while OB and BB will give rise to type B blood. Since the A and B alleles demonstrate codominance, having both the A and B alleles will give rise to type AB blood.
When a person doesn’t have a particular agglutinogen on the surface of his red blood cells, he develops the corresponding anti-agglutinogen antibodies which are known as agglutinins. For example, if my blood type were B, then I would develop anti-A agglutinins. If my blood type were O, then I would have both anti-A and anti-B agglutinins.
Qn 1: a) If my blood type were A, what agglutinins (if any) would I have in my plasma? b) If my blood type were AB, what agglutinins (if any) would I have in my plasma?
Earlier, I mentioned that when blood transfusions were first attempted, agglutination and hemolysis often occurred. How does this happen? Well, this is because agglutinins are immunoglobulins and they are either IgG (immunoglobulin G with 2 binding sites) or IgM (immunoglobulin M with 10 binding sites). Hence, a single agglutinin can bind 2 or more RBCs at the same time, thereby causing RBCs to be bound together by agglutinin. Such binding causes many RBCs to clump together, which is the process of ‘agglutination‘. These clumps then plug all the small blood vessels throughout the circulatory circulation. While stuck in these small vessels, the RBCs in these clumps either undergo physical distortion or phagocytotic attack which destroys the membranes of the agglutinated cells. This releases hemoglobin into the plasma, which is called hemolysis of the RBCs.
Qn 2: Apart from blood transfusion, how are agglutinins produced within the blood of people who do not have the corresponding agglutinogens on their RBCs?
Well, that was a lot of text. Secondly, there is also this thing called the Rh system. There are 6 common types of Rh antigens, each of which is called an Rh factor. They are C, D, E, c, d, e. A person with the C antigen will not have the c antigen, while a person with the c antigen will not have the C antigen. The same works for D-d and E-e. Because of the manner of inheritance of these factors, we all have one antigen from each of these 3 pairs of antigens. The type D antigen is widely prevalent in the population and is way more antigenic than the other Rh antigens. It is precisely the presence and absence of this type D antigen which is responsible for the (+) and (-) you see in blood group type labels respectively. So a person with antigen “D” would be Rh-positive, while a person with antigen “d” (i.e. does not have antigen “D”) would be Rh-negative.
Qn 3: We normally hear of people having (+), a Rh-positive, in their blood types way more frequently than (-), Rh-negative. From a simplified perspective, do you think that the gene that codes for antigen D is autosomal dominant, or do you think that it is autosomal recessive?
The major difference between the earlier discussed O-A-B system and the Rh system is that in the latter, spontaneous agglutinins almost never occur. Instead, the person must first be exposed to large amounts of the Rh antigen before enough agglutinins develop to cause a significant transfusion reaction.
Qn 4: Given the info in Qn 3 and your response to it, why do you think it can be problematic when a Rh-negative woman is pregnant? (Hint: her husband is likely to be Rh-positive; what does this mean for her fetus?)
Having given a pretty decent overview (hopefully) of the two main blood type systems, I want to share with you a piece of amazing news I read which prompted me to write this article in the first place. Just a few days ago, researchers analysing bacteria in the human gut published an article in Nature, a leading peer-reviewed journal, where they discussed how they discovered that microbes there produce two enzymes which can convert type A blood to type O blood. If all goes well, this could revolutionize blood donation and transfusion.
As we have discussed above, type O blood has no A or B agglutinogen, and this makes type O donor blood ideal as it is virtually universal. Scientists have tried transforming the second-most common blood type, type A, into type O by snipping off the terminal N-acetylgalactosamine from antigen A. Recall this excellent diagram:

This has had limited success because currently known enzymes which can catalyse the conversion of antigen A to antigen H are not efficient enough to make the process economically feasible.
After 4 years of trying to improve on those enzymes, a team led by Stephen Withers, a chemical biologist at the University of British Columbia (UBC) in Vancouver, Canada, decided to look for a more efficient enzyme produced by bacteria in the human gut. Some of these microbes latch onto the gut wall, where they digest the mucins. Mucins’ sugar groups are similar to that which are found on the antigens on RBCs.
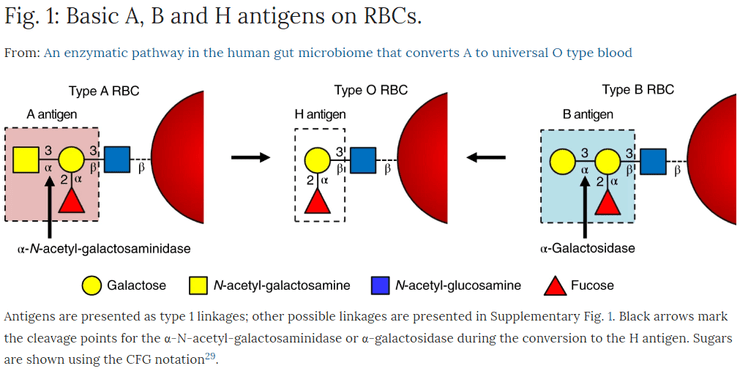
So, UBC postdoc Peter Rahfeld collected a human stool sample and isolated its DNA, which in theory would contain genes that encode the bacterial enzymes that digest mucins. Chopping this DNA up and loading different pieces into copies of the commonly used lab bacterium Escherichia coli, the researchers monitored whether any of the microbes subsequently produced proteins with the ability to transform agglutinogen A into agglutinogen H (its exclusive expression in the absence of antigens A and B defines blood type O).
At first, nothing promising was found. However, when two of the resulting enzymes from the obligate anaerobe Flavonifractor plautii were tested simultaneously, the N-acetylgalactosamine could be removed from antigen A. This worked because the two enzymes worked together in a pathway. First, N-acetylgalactosamine is deacetylated to galactosamine in a reaction catalyzed by N-acetylgalactosamine deacetylase.
With the second enzyme, the galactosamine linkage was cleaved. The cleavage would not have occurred unless antigen A was first deacetylated. The necessity of having both enzymes is illustrated in the image below:
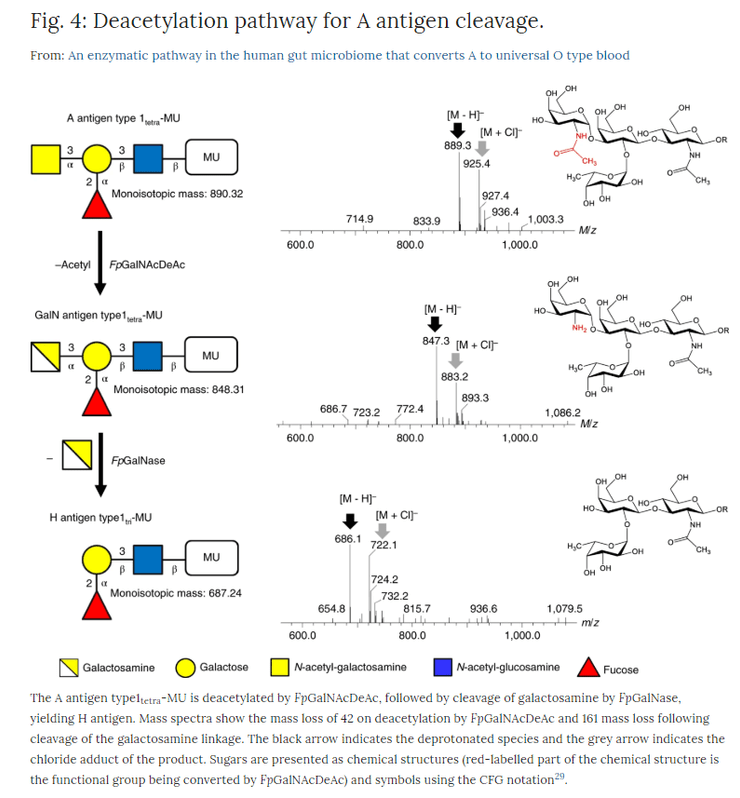
The most remarkable thing about this pair of enzymes is their ability to completely convert antigen A to antigen H on RBCs of the same rhesus type at very low enzyme concentrations. If this finding can be replicated, then this could indeed simplify their incorporation into blood transfusion practice, increasing the amount of “universal donor” blood.
Qn 5) Explain why people with the hh/Bombay blood type cannot receive supposedly “universal donor” type O blood.
Answers
Ans 1a) Anti-B agglutinins;
Ans 1b) Neither the anti-A nor the anti-B agglutinins
Ans 2) Small amounts of A and B agglutinogens enter the bloodstream via food, and via bacteria, and these agglutinogens initiate the development of the corresponding anti-A and anti-B agglutinins.
Ans 3) The gene that codes for antigen D on the surface of red blood cells is autosomal dominant.
Ans 4) The fetus is affected by the pathological Erythroblastosis Fetalis, aka: “Hemolytic Disease of the Newborn” (HDN). Since the gene that codes for Rh-positive is autosomal dominant, a Rh-negative woman is statistically more likely to have Rh-positive man than a Rh-negative man. This also means that the fetus is statistically more likely to Rh-positive. When the fetus is Rh-positive, the fetus has antigen D on its RBCs. The mother develops anti-D antibodies due to exposure to the fetus’ RBCs which express antigen D. Now, this is fine for the mother since she doesn’t have the antigen D. However, the synthesised anti-D agglutinins diffuse through the placenta into the fetus, and cause RBC agglutination. Good luck to the fetus. The agglutination leads to fetus RBC hemolysis and the released hemoglobin is broken down to bilirubin by the fetus’ macrophages. Bilirubin is a yellow substance and it is this that causes the baby’s skin to become yellow (jaundice). The anti-D agglutinins circulate for another 1-2 months after birth, destroying even more RBCs.
Ans 5) People with the Bombay blood type lack the terminal fucose group that is found on agglutinogen H. Hence, they have antigen h instead and thus produce anti-H agglutinins. When a person with Bombay blood receives type O donor blood, the anti-H agglutinins in the plasma will bind with antigen H found on the RBCs of type O blood. This will trigger the adverse blood transfusion reaction.
Hope you enjoyed this post!
Yours faithfully,
Nic Loh
12 Jun 2019
References
1. https://www.sciencemag.org/news/2019/06/type-blood-converted-universal-donor-blood-help-bacterial-enzymes
2. https://en.wiktionary.org/wiki/erythro-
3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4335469/
4. Unit 6, Chapter 36 of the 13th edition of Guyton-Hall Medical Physiology
5. https://en.wikiversity.org/wiki/Obstetrics_and_Gynecology/Rhesus_Disease
6. https://en.wikipedia.org/wiki/ABO_blood_group_system
7. https://biology.stackexchange.com/questions/44569/is-h-antigen-considered-as-an-agglutinogen
8. https://en.wikipedia.org/wiki/H_antigen
9. https://brainly.in/question/2534046
10. https://www.nature.com/articles/s41564-019-0469-7
11. https://en.wikipedia.org/wiki/N-Acetylgalactosamine

Leave a comment